Research
Welcome! Our lab is fascinated by the growth and morphogenesis of living matter: we try to understand how cells, multicellular colonies, and tissues execute their growth, and how they grow into precise, programmed shapes. Currently, we investigate several organisms, including bacteria, fungi, protists, and a plant! Investigating similar questions in diverse model and non-model systems within one lab helps us gain intuition for broad principles of growth and morphogenesis while also allowing us to identify the unique and beautiful strategies that specific organisms or taxa use to control these processes.
Regardless of the specific case, robustly executing growth and/or morphogenesis requires sophisticated control systems that include biochemical, biophysical, and genetic components. We therefore combine tools and and concepts from molecular microbiology, biochemistry, biophysics, and genetics to dissect these systems. We take an experiment-forward approach but often use physics-based modeling and theory to understand our data. Below are a few more notes on specific areas of interest within the lab. For details, please see publications or preprints, or feel free to contact us.

Bacterial Growth
Many bacteria are truly amazing in their capacity for rapid growth. We ask how single bacterial cells assemble, sculpt, and monitor the various layers of their envelope (plasma membrane, cell wall, and outer membrane) during their growth. Since the envelope determines cell shape, we are also inherently asking how cells maintain their highly precise shapes during growth. As part of this effort, we have developed several precise methods for measuring the physical properties of the cell envelope.
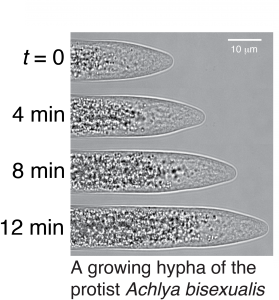
Hyphal Growth and Mycelial Development
Many fungi and protists grow as complex networks (mycelia) of interconnected tubes (hyphae). Each hypha grows from from a single pole, and the coordinated growth of all the hyphae in a mycelium leads to its growth. At the cellular level, we are studying how and why individual hyphae adopt specific shapes at the growing pole. At the mycelial level, we are studying how hyphae communicate with each other to coordinate their growth rates, and how this cell-cell communication leads to developmental stages of the mycelium.
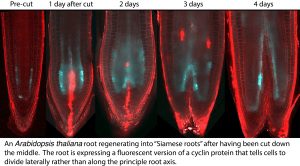
Plant Root Development and Regeneration
Plant roots are comprised of a limited number of cell types that are precisely patterned spatially. To achieve this, the proliferation of specific cell-types must be coordinated with tissue growth and global root shape. To understand the complex control system that executes this coordination we examine the remarkable ability of roots to regenerate this organization de novo after complete excision of its stem cell niche.